Zinc toxicity at high pH and low Ca concentrations and formulation of a generalized bioavailability model (gBAM) for D. magna
 Toxicity of metals like zinc (Zn) to freshwater organisms is not only dependent on the concentration of the metals itself, but also on other properties of the water, like its hardness and its acidity (pH). This is called bioavailability. In the European Union, safe levels of Zn can be calculated as a function of these properties, using so-called bioavailability models. Previously, the existing models could not be applied to more than 25% of European waters, because hardness or pH were higher than those for which the models were originally developed. In this research, we have shown with new experimental work that the existing model for algae can also be used at a much wider range of hardness and pH than previously thought. Our work with water fleas, however, showed that the existing model needed to be considerably improved. This improved model was called the generalized bioavailability model (gBAM) and is an important step to better toxicity predictions of zinc and more certainty in the calculation of environmentally safe concentrations of Zn in rivers and lakes.
Toxicity of metals like zinc (Zn) to freshwater organisms is not only dependent on the concentration of the metals itself, but also on other properties of the water, like its hardness and its acidity (pH). This is called bioavailability. In the European Union, safe levels of Zn can be calculated as a function of these properties, using so-called bioavailability models. Previously, the existing models could not be applied to more than 25% of European waters, because hardness or pH were higher than those for which the models were originally developed. In this research, we have shown with new experimental work that the existing model for algae can also be used at a much wider range of hardness and pH than previously thought. Our work with water fleas, however, showed that the existing model needed to be considerably improved. This improved model was called the generalized bioavailability model (gBAM) and is an important step to better toxicity predictions of zinc and more certainty in the calculation of environmentally safe concentrations of Zn in rivers and lakes.

Scientific Abstract
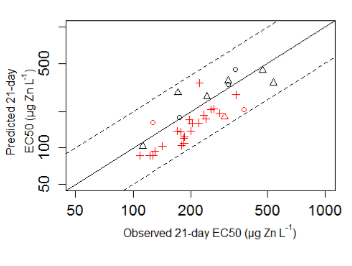
Risk assessment in the European Union implements Zn bioavailability models to derive predicted no effect concentrations (PNEC) for Zn. These models are validated within certain ‘boundaries’, i.e. pH≤8 and Ca concentrations ≥ 5mg/L, but a substantial fraction of the European surface waters falls outside these boundaries. Therefore, we evaluated if the chronic Zn Biotic Ligand Model (BLM) for Daphnia magna and the chronic bioavailability model for Pseudokirchneriella subcapitata, can be extrapolated to pH above 8 and Ca concentrations below 5mg/L. Results from D. magna experiments suggested that the BLM is not able to reflect the pH effect over a broad pH range (5.5-8.5). In addition, due to calcium deficiency of D. magna in the softwater tests, we can't conclude whether the BLM is applicable below its Ca boundary. Results for P. subcapitata experiments showed that the bioavailability model can accurately predict Zn toxicity for Ca concentrations down to 0.8mg/L and pH values up to 8.5. Because the chronic Zn BLM for D. magna could not be extrapolated beyond its validity boundaries for pH, a generalized BioAvailablity Model (gBAM) was developed. Of 4 gBAMs developed, we recommend the use of model gBAM-D, which combines a log-linear relation between the 21-d median effective concentrations (expressed as free Zn2+ ion activity) and pH, with more conventional BLM-type competition constants for sodium, calcium and magnesium. This model is a first step in further improving the accuracy of chronic toxicity predictions of zinc as a function of water chemistry, which can decrease the uncertainty in implementing the bioavailability based PNEC in the risk assessment of high pH and low Ca concentration regions in Europe.
Full reference (link)
Van Regenmortel T, Berteloot O, Janssen CR, De Schamphelaere KAC. 2017. Zinc toxicity at high pH and low Ca concentrations and formulation of a generalized bioavailability model (gBAM) for D. magna. Environmental Toxicology and Chemistry (Accepted Article).
